Caring for someone with PWS?
This remote study is designed to better understand caregiver impact and well-being by capturing everyday experiences of those caring for individuals with PWS on the Folia app.
Interested? Let’s see if this study is a match
Download the Folia app and use the invite code “BRAVE” to sign up or learn how to enroll here.

Why tracking matters for PWS?
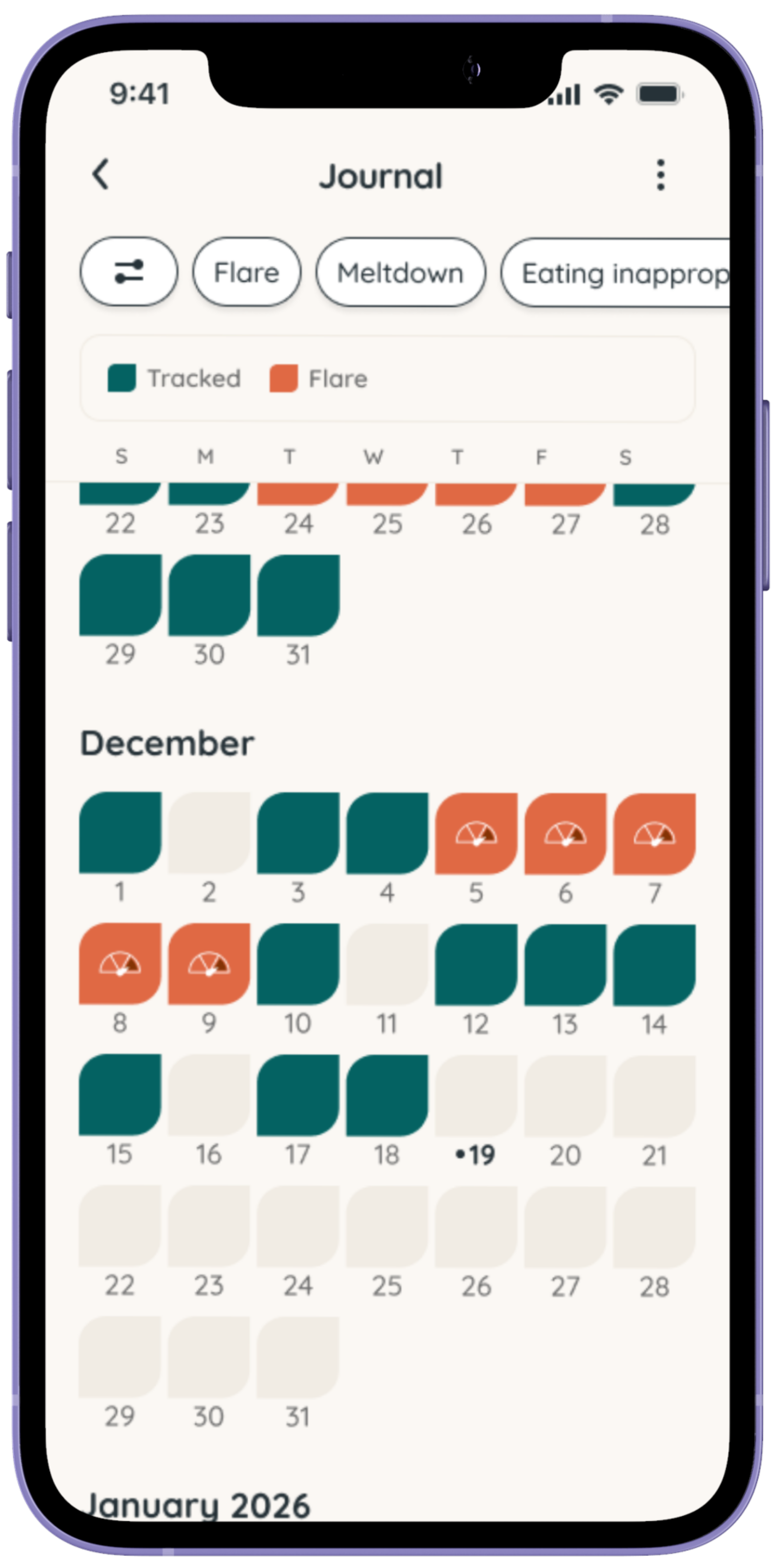
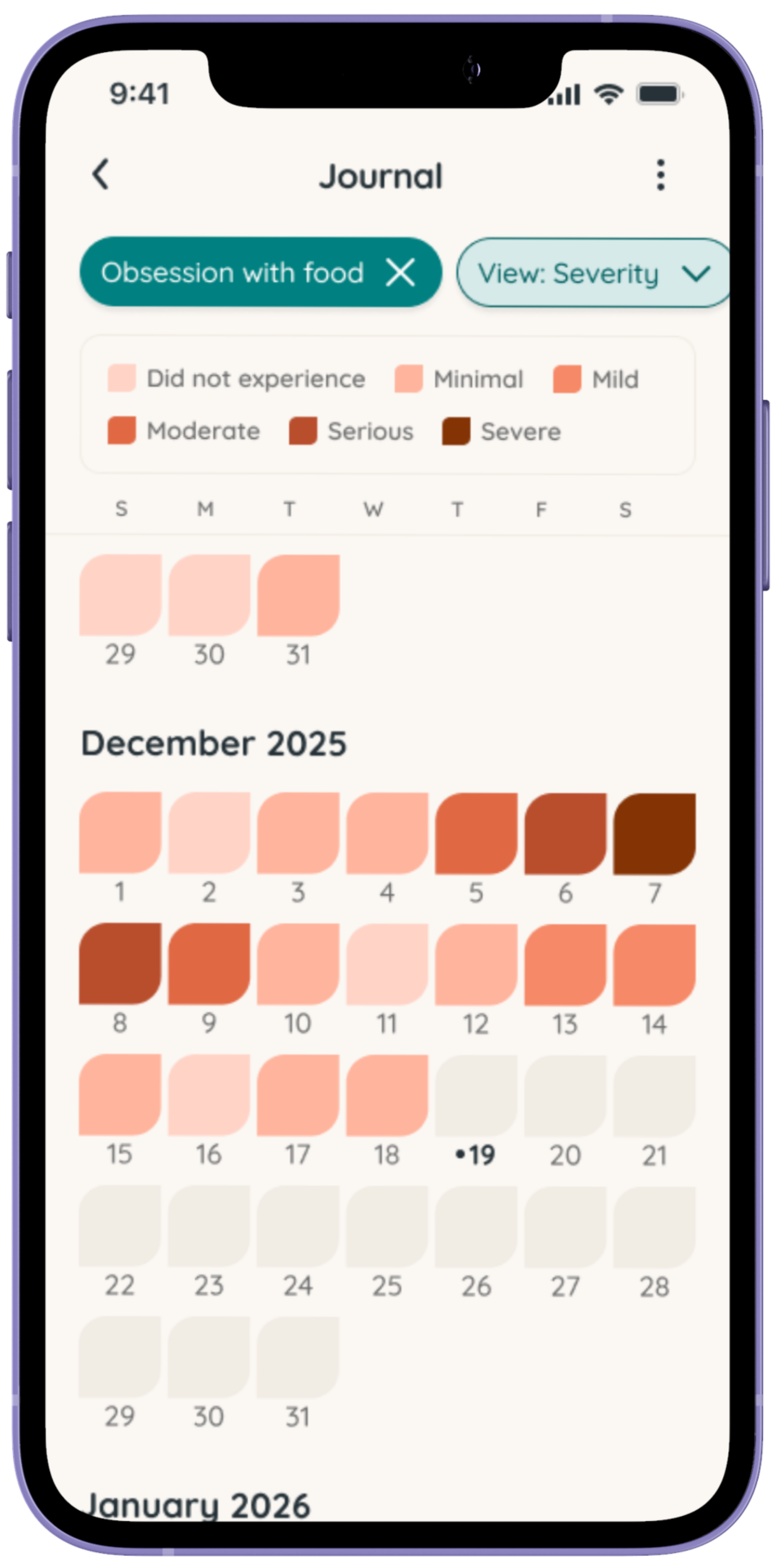
Tracking can help bring clarity to the day-to-day experience of living with Prader-Willi Syndrome (PWS). Symptoms like hyperphagia, behavior changes, sleep challenges, and responses to routines or treatments can vary, and patterns are often hard to spot in the moment.
Using Folia allows caregivers of individuals with PWS to log observations, symptoms, and treatments over time. This can help families notice trends, prepare for medical appointments, and better understand what may influence hunger, behavior, and overall wellbeing.
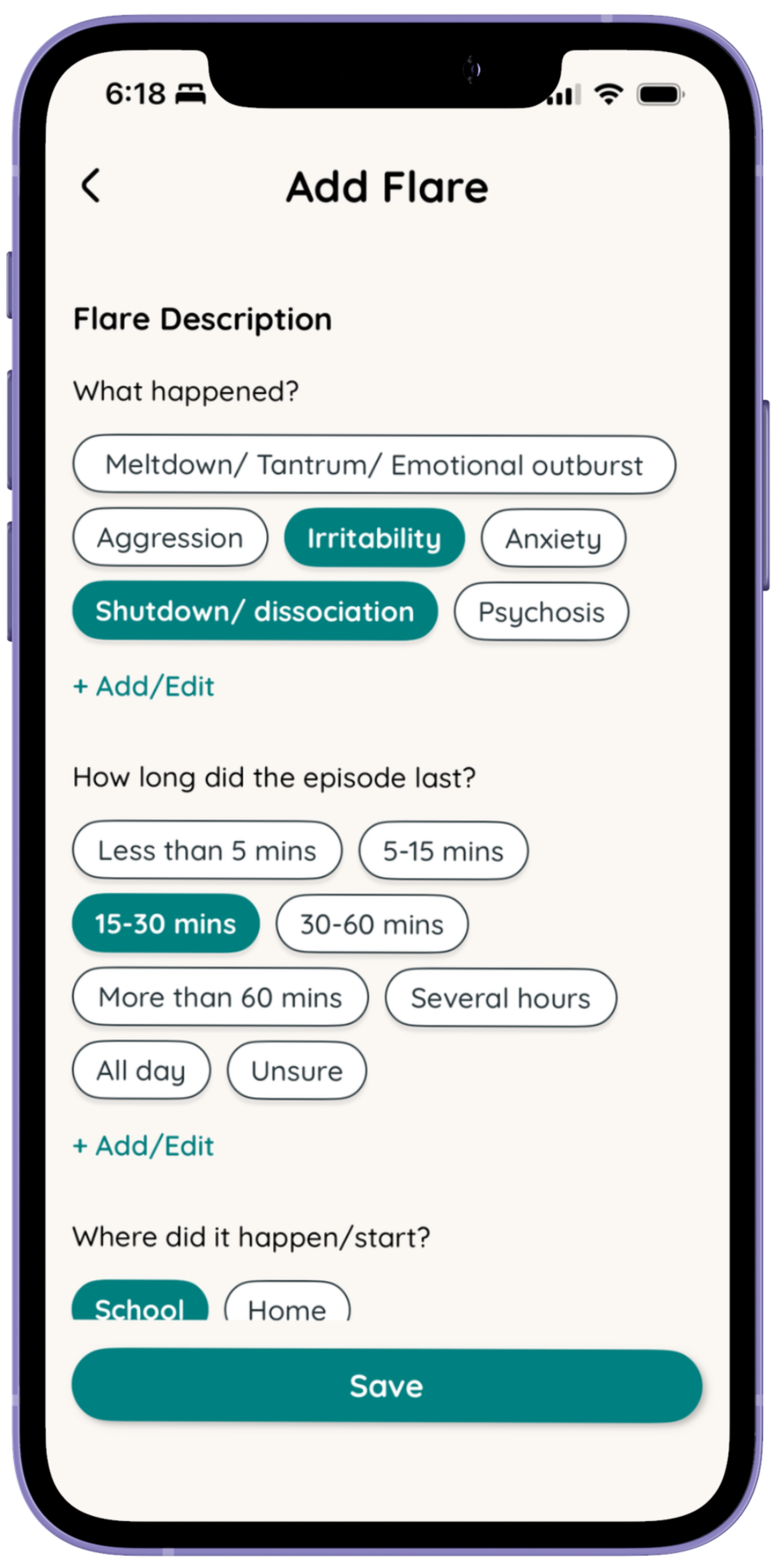
How it works
While static trackers and discussion guides exist , Folia is the first and only interactive tool that captures, visualizes, and lets individual and their caregivers reflect on their own longitudinal PWS experience.
We recognize the role caregivers play every day, tracking on Folia helps capture that.
Share weekly updates
Both for yourself as the caregiver and for the individual with PWS
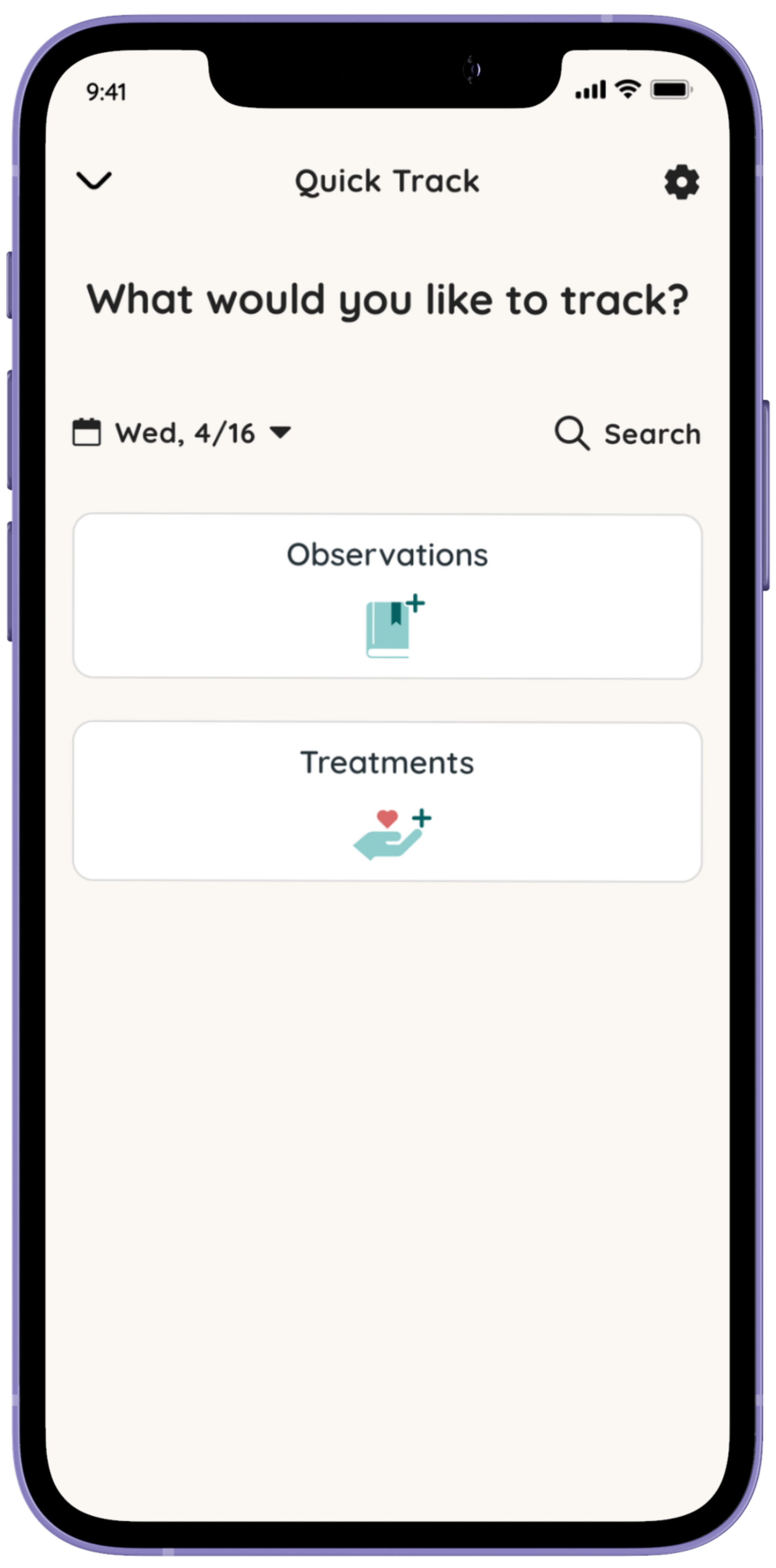
See insights about the impact of your health as a caregiver.
Respond to short monthly check-ins in the Folia app
Participants will be compensated for their time and involvement in the study.
What is PWS?
Prader-Willi Syndrome (PWS) is a rare neurodevelopmental disorder caused by abnormal gene expression on chromosome 15. This high-impact condition affects approximately 1 in every 15,000 live births. PWS is associated with hypotonia, cognitive impairment, and hyperphagia.
Hyperphagia, a defining feature of PWS, is an intense and persistent sensation of hunger and preoccupation with food that often requires lifelong, 24/7 supervision from family members or caregivers. This level of care can significantly impact the quality of life for both individuals with PWS and their caregivers.
Why are we researching PWS?
The goal of the BRAVE-PWS Caregiver Study (Burden and Real‑world Assessment of caregiver Voice and Experiences in PWS) is a Soleno Sponsored Study to better understand and characterize the everyday experience of PWS caregivers. As new treatments are developed, it’s important to understand their effects not just on the individual with PWS but also on their support system.
Interested in joining?
Caring for someone with PWS?
To participate in this study, all of the following must apply to you:
You are the primary caregiver for an individual age 4 or older who has been diagnosed with Prader-Willi Syndrome (PWS).
The individual with PWS must not be taking VYKAT™ XR (diazoxide choline) extended-release tablets
You live in the United States.
You are at least 18 years old (or the age of majority in your state).
You are able to read and understand English.
Diagnosis of PWS will be confirmed through self-reported screening procedures and patient- or caregiver-supplied documentation. Confirmation of diagnosis for each participant will be reviewed by the Folia Health study team as part of standard validation procedures.

How to enroll
Review the eligibility criteria.
If the study looks like a fit, download the Folia Health app.
Use invite code “BRAVE” at signup to ensure you’re enrolled into the study
Review and sign the consent form (you’ll see it right after you create your account)
Reach out to [email protected] with any questions!